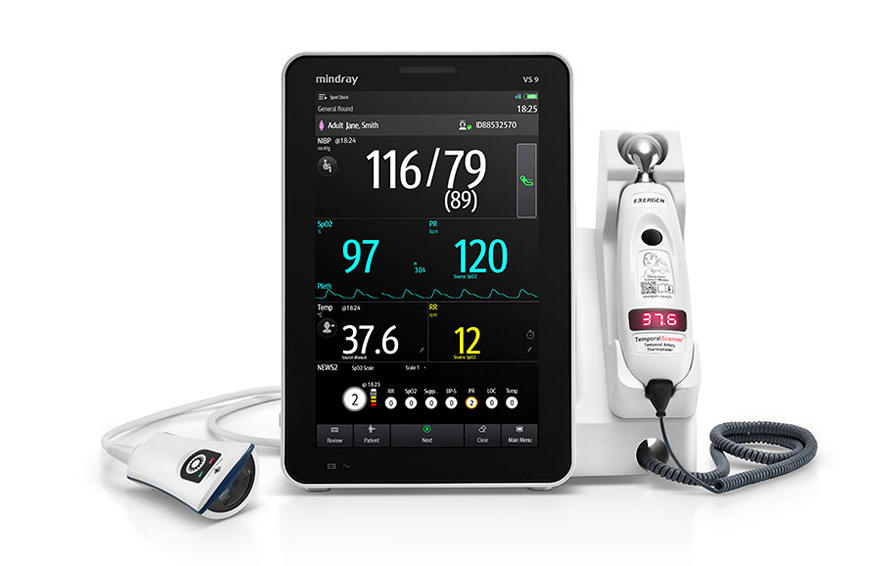
Featuring innovative technology and connectivity to help clinicians get a complete picture of patient health, Mindray’s new VS9 Vital Signs Monitor is slated to elevate clinical workflow.
Mindray, a leading global developer of healthcare technologies and solutions for ultrasound, patient monitoring, and anesthesia, announced the launch of their new vital signs monitor, the VS9. Loaded with functionality and parameters that provide tools to support clinical decisions, the VS9 offers spot check and continuous vital signs monitoring to help clinicians get a complete picture of patient health. The VS9 Vital Signs Monitor features a 10.1″ touchscreen display and many intuitive tools to enable clinicians to streamline clinical workflow, reduce input errors, improve patient outcomes, and enhance patient satisfaction.
The VS9 Vital Signs Monitor helps clinicians elevate safety, provide true interoperability, and ensure reliability.
Elevate Patient Safety
- True to life parameters include non-invasive blood pressure, pulse oximetry, and versatile temperature measurements
- Improve patient outcomes with the addition of CO2 for continuous monitoring for early detection of respiratory depression
- Validate patient information and store up to 5,000 measurements, accessible by patient ID for easy review
- Standard Modified Early Warning Score (MEWS) offers support for clinical decisions
Provide True Interoperability
- Improve clinical workflow with configurable workflows defined by department
- Streamline department workflow and improve mobility between patients by sending vital signs at the point of care directly to the EHR
- Centrally monitor patients with connectivity to the BeneVision Distributed Monitoring System
Offer True Quality and Reliability
- Manufactured with durable materials to withstand high-volume medical environments
- Built and tested to protect against device deterioration due to chemical exposure
“At Mindray, we understand that the emphasis on patient and clinician safety has never been higher,” said Wayne Quinn, President of Mindray North America. Quinn continued, “Our mission to advance medical technologies to make healthcare more accessible is relevant now more than ever. We are excited to introduce innovative technologies and connectivity tools with the VS9 Vital Signs Monitor to help clinicians maximize patient safety, improve patient outcomes, and increase efficiency in the Med-Surg Unit and beyond.”
The easy-to-use VS9 Vital Signs Monitor offers functionality and parameters to support clinical decisions and elevate your department’s workflow.
V9 Vital Signs Monitor Features and Benefits
- TrueBP™ quickly measures blood pressure on inflation in about 15 seconds
- BP Averaging functionality provides an average BP over a specified time and a number of measurements
- Orthostatic BP helps assess a patient’s fall risk
- Customize up to 30 customizable manual inputs, such as respiration rate, glucose, and height
- Manual, automatic, and customizable NIBP measurement modes offer flexibility for various clinical applications
- Hardwired or wireless configurations available with Bluetooth connectivity
With over 40 years of vital signs monitoring experience, Mindray understands how to deliver clinician-centric, high-value solutions. They offer the industry’s best total cost of ownership, including an industry-leading 3-year warranty for all vital signs products, low acquisition costs, and the ability to deploy on a hospital’s existing network. Through innovative solutions like the VS9 Vital Signs Monitor, Mindray continues to drive its mission forward, advancing medical technologies to make healthcare more accessible.
About Mindray
Mindray is a leading developer, manufacturer, and supplier of medical device solutions and technologies used in healthcare facilities around the globe. We believe we can change lives by making the most advanced healthcare technology attainable for all. We do this by empowering healthcare professionals through innovative, high-value solutions that help create the next generation of life-saving tools across three primary business segments: patient monitoring and life support, in-vitro diagnostics, and medical imaging. Mindray maintains its global headquarters in Shenzhen, China; Mindray North America is headquartered in Mahwah, New Jersey. Our Ultrasound Innovation Center is located in San Jose, California with additional facilities in major international markets around the world. For more information, please visit https://www.mindray.com.